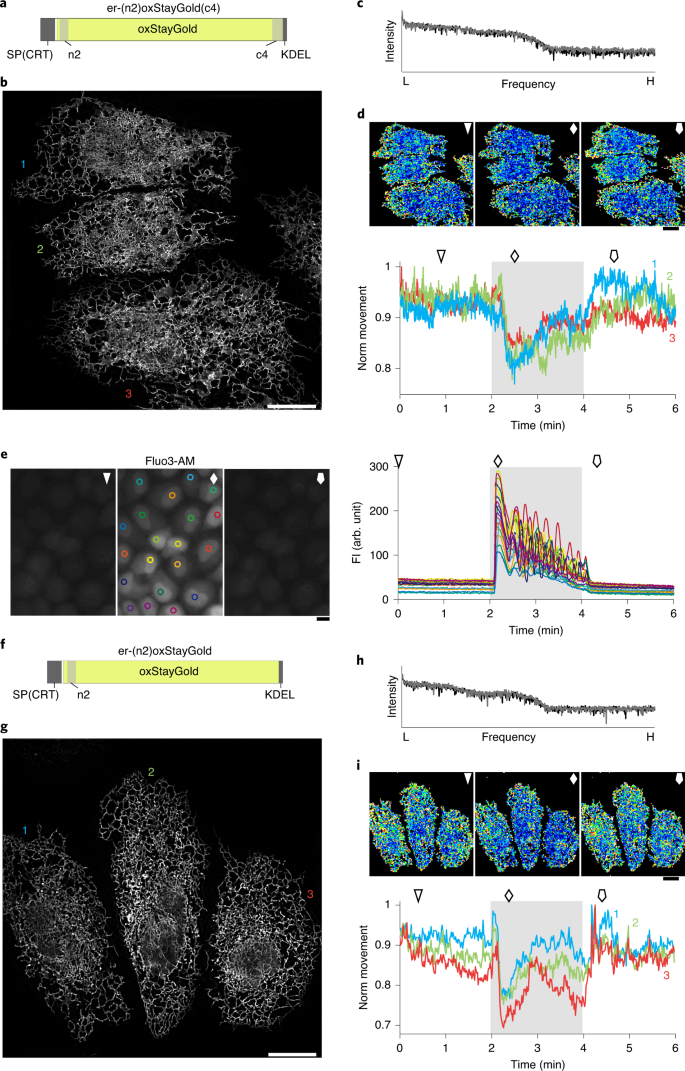
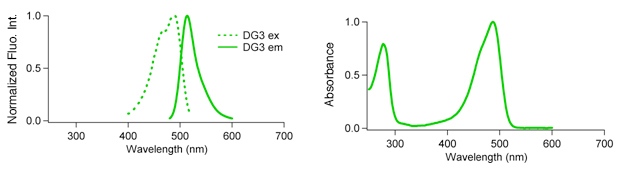
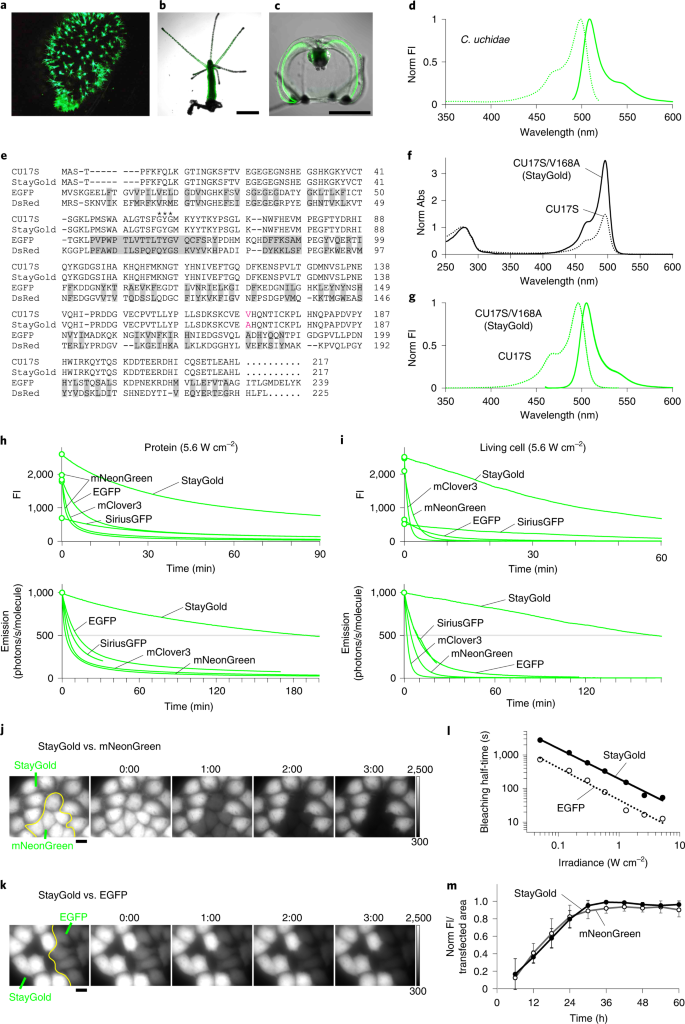
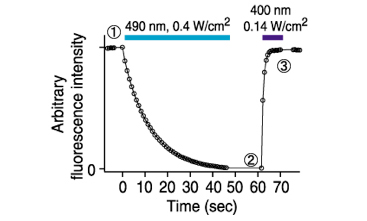
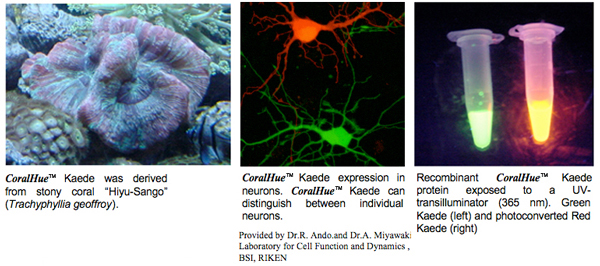
An optical marker based on the UV-induced green-to-red photoconversion of a fluorescent protein | PNAS
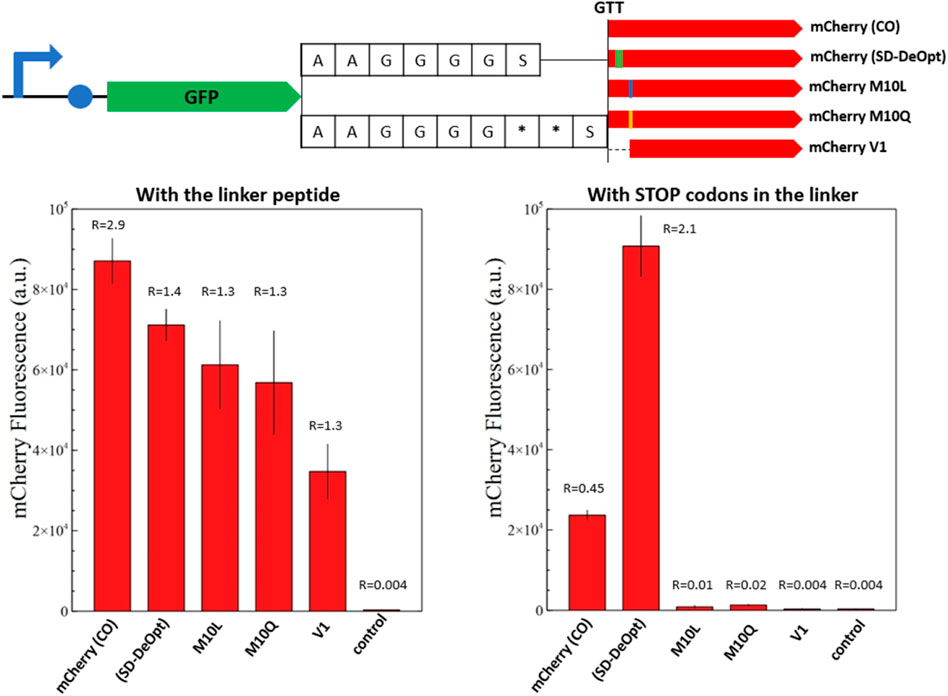
Frontiers | mCherry contains a fluorescent protein isoform that interferes with its reporter function

Ultrafast Excited-State Dynamics of the Photoswitchable Protein Dronpa | Journal of the American Chemical Society

Acid-Tolerant Reversibly Switchable Green Fluorescent Protein for Super-resolution Imaging under Acidic Conditions - ScienceDirect

Low-Temperature Chromophore Isomerization Reveals the Photoswitching Mechanism of the Fluorescent Protein Padron | Journal of the American Chemical Society

IJMS | Free Full-Text | Photoconvertible Fluorescent Proteins and the Role of Dynamics in Protein Evolution

Absolute quantum yield measurements of fluorescent proteins using a plasmonic nanocavity | Communications Biology